
Written by: Steven Jiang, MSc Student, Kim Lab (pictured)
Researchers at the UBC Centre for Blood Research (CBR) have made a significant advancement in how to check the quality of red blood cells for transfusion. They identified a practical and efficient method to help assess the quality of red blood cells (RBCs) in stored bags, without wasting bags during the testing process. Emel Islamzada, the first author of a recent paper on this subject and a PhD Candidate in UBC’s Multi-Scale Design Laboratory, talked to us about her work.
During blood donation, a donor’s blood is captured in two different areas: the blood bag and a length of tubing called the segment (Figure 1). The blood from the bag will be transfused into a patient while the blood from the segment will not. To check a blood bag’s quality, it’s possible to directly sample blood from the bag, but this method can waste or contaminate the entire unit. Instead, healthcare providers could sample blood from the bag’s segment to assess quality. However, this comes with another problem: RBCs degrade at different rates in segments and in bags, so it was unknown if the blood in these two areas had similar properties.
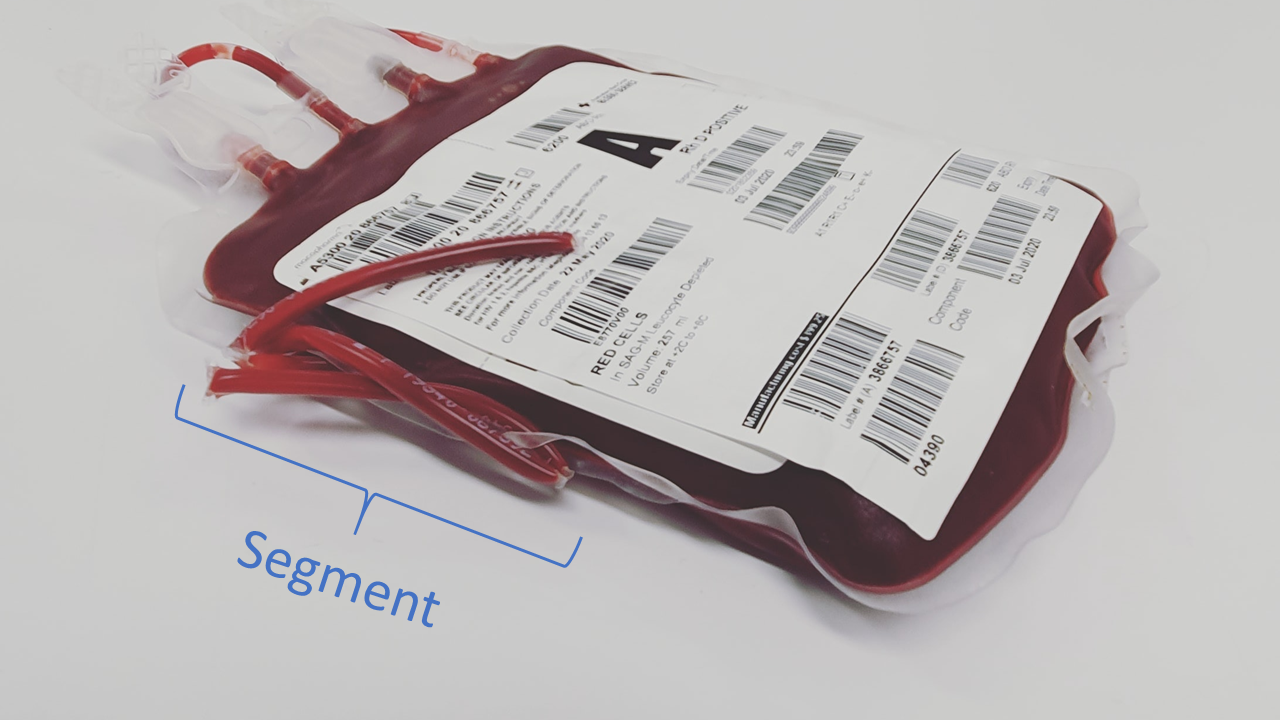
Figure 1. An RBC blood bag, with segment tubing labeled. Image via Pexels.
This is where Emel and her colleagues stepped in. In their recent study published in Transfusion, they tried to confirm if the deformability of blood in the segment was similar to the properties of blood in the bag. If so, a viable, non-wasteful option to test for blood quality could be simply to sample blood from the segment.
To assess blood quality in both the bag and segment, they looked at a biomarker called deformability. Deformability is a red blood cell’s ability to squeeze and change shape – crucial for its job, where it must squeeze through tiny blood vessels while delivering oxygen throughout the body. The longer that donated RBCs are stored, the less deformable and more rigid they become, making them less useful to the transfusion recipient.
An RBC’s rigidity, or lack of deformability, could therefore be useful in assessing the quality of donated RBCs. Emel argues that RBC rigidity has been used as a highly sensitive biomarker for malaria infection, which contributed to her lab’s idea to measure it in this study.
The device used to test rigidity, called a microfluidic ratchet, was not commercially available (Figure 2). To tackle this issue, the engineers in Emel’s lab designed this device, and Emel was responsible for making them. She says, “I’ve been doing a lot of the bench work. Our devices are single-use, so I make them all from scratch. Then, I run the assays and analyze the data.”
Using this device, Emel tested and compared the deformability of the RBCs from the blood bag and the segment every 2 weeks up to 8 weeks. She found that the RBCs from the blood bag and the segment have similar deformability across all samples and all timepoints.
Through their work, the researchers confirmed that the deformability of RBCs are similar in both the segment and bag over time2, suggesting that it’s sufficient to sample from segments to check blood bag quality.

Figure 2. The microfluidic device used for rigidity assay. The image was captured under a microscope. The figure was adapted from Islamzada et al., 20211
Despite the major engineering breakthrough, more research is still required before Emel and her colleagues can bring their product into clinics. They are unsure if the rigidity of the RBCs is an accurate representation of the overall quality of these RBCs in human. Animal and clinical studies are currently ongoing to answer this question. However, Emel is optimistic, and she expects that the rigidity test will be used in addition to current biochemical tests in the future, which could help healthcare providers to transfuse the RBCs of the best quality to sensitive transfusion patients.
We asked Emel about how she and her colleagues felt when this work was published. She says, “We are quite proud to contribute to this research area. There are still burning questions for us to answer, and we still need reliable, robust, and relatively easy ways to check for the quality of the red blood cells. But when you look at the amount of work that has been done to ensure that transfusions are as safe and effective as possible, it’s very humbling to be a part of it.”
References
- Islamzada, E., Matthews, K., Guo, Q., Santoso, A. T., Duffy, S. P., Scott, M. D., & Ma, H. (2020). Deformability based sorting of stored red blood cells reveals donor-dependent aging curves. Lab on a Chip, 2(2), 226-235. https://doi.org/10.1039/c9lc01058k
- Islamzada, E., Matthews, K., Lamoureux, E., Duffy, S. P., Scott, M. D., & Ma, H. (2022). Blood unit segments accurately represent the biophysical properties of red blood cells in blood bags but not hemolysis. Transfusion (Philadelphia, Pa.), 62(2), 448-456. https://doi.org/10.1111/trf.16757


