By Mike Hughes, Research Associate, McNagny Lab, CBR
By Mike Hughes, Research Associate, McNagny Lab, CBR
Vampire folklore is perhaps one of the most ancient monster myths. Vampires are also culturally diverse, having been described by disparate cultures throughout history. Some scientists and clinicians have suggested that the global and culturally similar vampire descriptions may be explained by rare genetic blood disorders (e.g. porphyria), vitamin deficiency (e.g. pellagra) or infections (e.g. rabies). Of course, there is no medical explanation that accounts for all vampire powers- the pathophysiology of human-to-bat transformation has yet to be discovered! Rabies or a similar viral vector theory seems to appeal to modern storytellers perhaps because of a more science-savvy audience that understands viral transmission or because it is a convenient way to generate more vampires to pump up the dramatic tension.
In the Blade movie series, vampirism is caused by a parvovirus – a family of single-stranded DNA viruses – transmitted by saliva through a bite. Infection slows aging, provides superhuman strength and rapid healing, but also gradually destroys hemoglobin (the iron-containing protein in red blood cells responsible for carrying oxygen). The slow and steady loss of hemoglobin causes the insatiable thirst for human blood. In the Blade universe, vampires have a number of the usual weaknesses described in modern folklore, including aversion to sunlight (technically just the UV part of the spectrum) and a severe allergy to silver and garlic. However, Blade vampires have also a unique, violent reaction to ethylenediaminetetraacetic acid (EDTA): they explode upon contact.
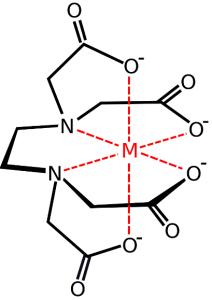
EDTA is a small molecule, that can be likened to a “six-fanged vampire” (the technical term is “hexadentate”), that grabs and holds on to metal cations like calcium, iron and zinc – a process called chelation. Unlike the dull, colorless bottle of EDTA that is collecting dust on every laboratory bench, in Blade, EDTA is an eye-catching cerulean-blue elixir loaded into bullet-shaped syringes.
Why EDTA exactly? The Hollywood hand waving goes something like this: EDTA is used clinically as a blood thinner and clot buster and vampires have sensitive hemoglobin (because of the virus, remember)… therefore, boom! I cannot comment on the latter part of the theory but EDTA chelation is used clinically to treat poisoning by lead and other heavy metals, emergency treatment of hypercalcemia (high calcium levels) and irregular heartbeat due to digitalis toxicity. In the body, EDTA-bound/chelated metals are more rapidly excreted in urine.
However, EDTA is most famous for its contentious use in an alternative treatment called “chelation therapy”. In this type of chelation therapy, patients receive an intravenous infusion of an EDTA solution. Treatments are usually given once per week for 20-40 weeks followed by “maintenance” treatments at longer intervals. Since EDTA can also deplete essential trace metals like iron, copper, manganese, zinc and others, the regimen usually includes high-dose mineral and vitamin supplements. The primary ailment(s) for patients seeking EDTA chelation therapy (ECT) is atherosclerosis-associated cardiovascular disease(s).
The purported rationale is that, EDTA chelates calcium (Ca2+) that results in a transient hypocalcemia (low calcium levels), which, in turn, decalcifies* and dissolves atherosclerotic plaques… therefore, cure! Admittedly, this seems more plausible than vampire-related applications for EDTA; however, neither claim has been tested in large-scale clinical trials.
Scientists and physicians have long refuted the clinical benefit of EDTA chelation therapy (ECT) for atherosclerosis and a host of other ailments (1). In 1989, the US Food and Drug Administration (FDA) included ECT on its list of “Top 10 Health Frauds”, although it was quickly removed for mysterious reasons. Furthermore, the safety of these treatments, administered as intravenous solutions of various (and sometimes secret components), is questionable (1, 2).
Although rare, deaths from cardiac arrest due to hypocalcemia during ECT have been reported (2). The ECT may do the most harm in patients that forgo effective medicines and treatments (including bypass surgery) in favor of ECT. Proponents of ECT would argue that, because there is no money in EDTA as a drug, clinicians and the pharmaceutical industry have denied its efficacy and blocked approval. However, in the United States, over 100,000 people received ECT in 2007 (up 68% from estimates in 2000) (3) and demand in Canada is probably very similar – you don’t have to look very hard to find a clinic offering ECT in British Columbia. ECT is primarily offered by alternative medicine practitioners at a cost of $5,000-10,000 for a typical course (including the mineral supplements). These fees are not typically covered by medical insurance, private or public.
A patient-led advocacy for off-label EDTA treatment was apparently so great that the US National Institutes of Health (NIH) commissioned a 5-year, $30 million clinical trial, called TACT (Trail to Assess Chelation Therapy), to test the efficacy of EDTA. The study enrolled 1,708 stable patients that had suffered myocardial infarction (heart attack) (3). However, the design and methods of the TACT study have been criticized for several reasons (2). Although the TACT study was designed to be randomized and placebo-controlled, the trial was conducted in part by “chelation-therapy practitioners” at “chelation therapy clinics” (2)†. Some of the TACT trial was conducted at naturopath clinics in Canada, including in British Columbia. Furthermore, besides EDTA, the ECT solution contained also unfractionated heparin (a proven blood thinner), ascorbic acid (vitamin C), pantothenic acid (vitamin B5), and an anesthetic (procaine) plus a few other ingredients‡. The placebo? It was just normal saline with 1.2% dextrose (4).
Although the chelation therapy did not differ from placebo in most individual experimental end points, there was a modest benefit for ECT patients in primary endpoints when combined (coronary revascularization, hospitalization due to angina, and non-fatal stroke or heart attack) (3). The benefit was largely restricted to patients with diabetes (5). Thus, the results of the trial, published in the March 2013 issue of the influential Journal of the American Medical Association (3), failed to put a stake through the heart of chelation therapy. The TACT investigators caution that their results do not endorse the widespread application of EDTA therapy for post-MI patients; however, some authors, including those involved in the TACT study, have published more optimistic follow-up (re)-appraisals that suggest diabetic patients might indeed benefit (6, 7, 8).
For better or for worse, the results of the TACT study have legitimized the claims of ECT proponents – most of whom still remain on the fringe of evidence-based medicine. Hopefully, the efficacy and safety of ECT for post-MI diabetic patients will receive continued scrutiny in future studies. Unfortunately, these results will also fuel the more dubious claims of chelation therapy clinics: ECT has been proposed to prevent or treat autism, Alzheimer’s disease, hypertension, cancer and atherosclerosis. The mechanism of action for these are various but typically involves the chelation (and excretion) of metals that catalyze formation of reactive oxygen species (ROS). Diminishing ROS, in turn, attenuates or reverses disease processes. Plausible? Yes, but evidence is required to support these claims. It is unlikely that another $30 million study will be conducted any time soon, leaving ECT in the realm of the scientifically undead – somewhere between quackery and legit.
* It seems the decalcification theory might be a bit passé in ECT circles. Other mechanisms have been proposed but none have been tested in animal models or human trials. See ref (2) for details.
† About 2/3 of the trials were conducted at ECT clinics throughout the US and Canada. The TACT findings suggest that this did not results in a measurable bias compared to data collected elsewhere. See Fig 3 in ref (3).
‡ TACT investigators were attempting to replicate the ECT methodology offered by ECT practitioners who also include these other ingredients. I could not find any explanation of the choice of placebo.